Active Sensation
(Science, my boy, is made up of mistakes, but they are mistakes which it is useful to make, because they lead little by little to the truth.*)
Overview
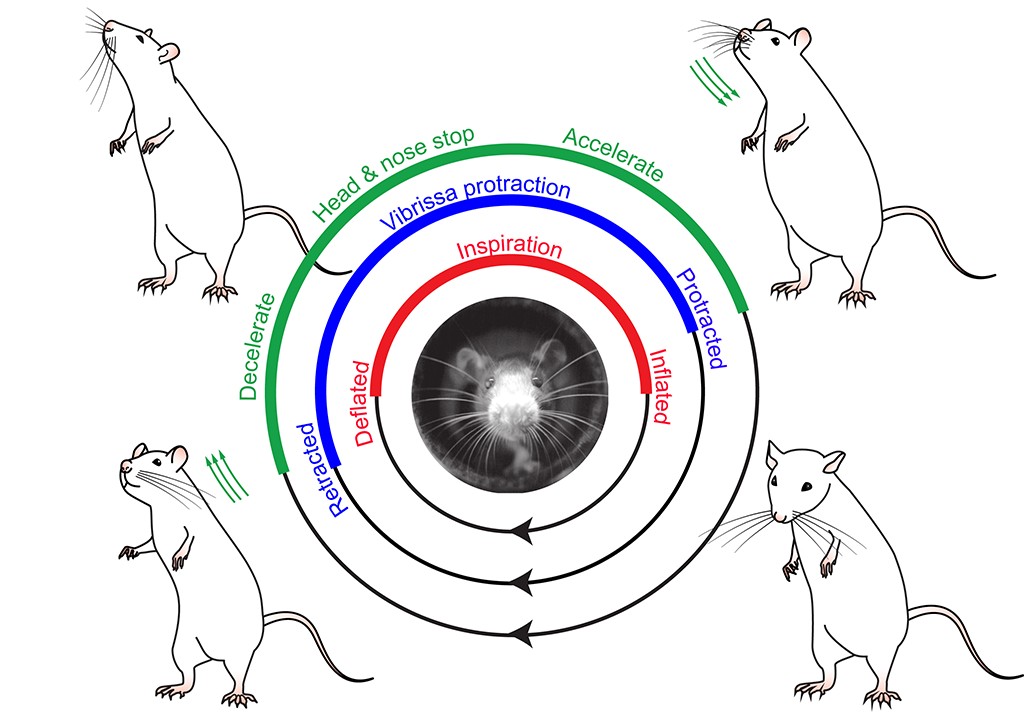
We consider how animals extract a stable picture of the world from the blur of inputs obtained with their actively moving sensors. The underlying computational problem is to combine a reafferent signal of sensor position with an exafferent signal of touch so that the animals can probe objects, identify their location in a body-centered coordinate system, and plan future actions. We work with orofacial behaviors, with aonecurrent focus on head movement and the vibrissa sensorimotor system of rats and mice; these animals palpate the world in front of them as they navigate and identify objects (top figure).
Signal Flow
The anatomy of the vibrissa sensorimotor system consists of a hierarchy of nested loops that support coherent rhythmic electrical signaling (middle figure). Feedback within the brain stem loop modulates the motion of the vibrissa and gates sensory transmission. Loops at the thalamic and neocortical levels appear to control vibrissa motion based on sensory input. The integration of control signals from inputs at multiple levels in the hierarchy is mediated by motoneurons in the facial nucleus. This stands as a model for deciphering, in general, the algorithm that underlies motor control of sensory input.
Ongoing Projects
Ongoing projects address: (i) The motor control network for head movement; (ii) Modulation of brainstem processing by feedback loops; (iii) Thalamic encoding and cortical computation of the coordinates of vibrissa touch; and (iv) Deep imaging for functional recording from brainstem nuclei.